“As Principal Deputy Commissioner, Namandjé will work closely with FDA leadership to develop, advance and implement key public health initiatives, as well as oversee the agency’s day-to-day functions,” FDA Commissioner Robert Califf told FDA staff in an email sent late last month. “Chief among those priorities is the proposed reorganization unifying the Human Foods Program, creating a new model for the Office of Regulatory Affairs, and strengthening the entire agency."
In this capacity, Bumpus will work closely with Jim Jones, who was recently appointed deputy commissioner for the Human Foods Program, which was proposed by the agency at the behest of the Reagan-Udall Foundation following a scathing audit of FDA’s botched response to an infant formula recall that resulted in massive national shortages.
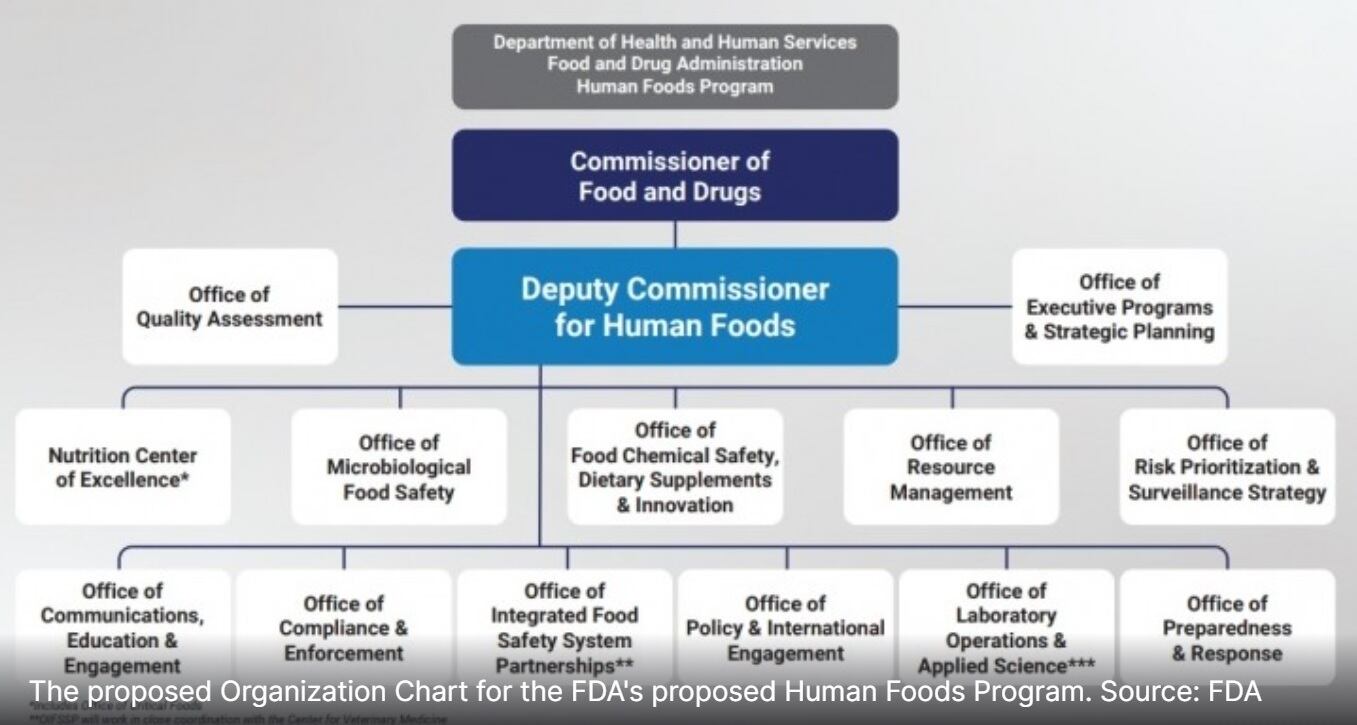
While the Human Foods Program likely will not be officially finalized until next summer, Jones is already working with leadership and field staff to advance an ambitious agenda centered on preventing foodborne illness, improving nutrition and enhancing chemical safety.
An advocate for ‘truth-telling in public health’ to combat misinformation
A central theme in Jones’ plan for the Human Foods Program is an increased emphasis on clear communication and enhanced transparency – values that Califf says Bumpus also shares.
He explained that in her role as chief scientist, which she took in August 2022, Bumpus “has continued to raise the cache of the FDA’s regulatory science within the agency and to the outside world, in part by being a champion of plain language, a staunch advocate for truth-telling in public health, and a formidable scientist.”
Also in her current role, Bumpus has actively fought against misinformation – which is an emerging challenge within public discussions of food safety. For example, differences of opinion contributed to recent additive bans passed in California, which Jones has said he would like to “get in front” of.
For her part, Bumpus told the Alliance for a Stronger FDA earlier this year that to address misinformation she is “very interested in getting out scientific data,” in a way that people with different educations and experiences can understand.
“When we’re talking about misinformation and people looking at data, one thing that’s become clear to me is that there is a piece of it that is differences in interpretation due to people having different levels of familiarity and comfort with even looking at data. So, some people look at data all the time, and they’re very comfortable, they know what it means. Other people don’t, and it’s hard to interpret. And that means they’re very reliant on interpretations from other people and what’s coming in around them.
“I think that there’s a real opportunity for FDA to lead and the scientific community to come together to provide more literacy around data and data analysis, and get getting people in general more comfortable with looking at data and understanding it,” she said.
While she was speaking directly about her current purview, as chief scientist, she added these are values she has held throughout her career.
She told the Alliance for a Stronger FDA this summer that when she was at Johns Hopkins University School of Medicine where she served as the E.K. Marshall and Thomas H. Maren Professor and chair of the Department of Pharmacology and Molecular Sciences she “used to do a lot of things in the community – community calls to talk about specific scientific topics” or visiting coffee shops to talk about her work and strategies related to HIV prevention.
“I think we can do more of that as a sign of community, and even FDA, trying to get the information out there,” she added, while simultaneously acknowledging existing efforts to educate the public through videos and existing outreach.
Jones also appears to share this value given his commitment to go into the field at least once a month.
Track record of collaboration could help repair trust in the new human foods group
Bumpus also has a track record of celebrating teamwork and collaboration in her current role, which would translate well to the development of the proposed unified Human Foods Program.
Many of the divisions of FDA that Jones will help bring together in his role have a history of acrimony, internal competition and distrust, which stemmed in part from the previous structure with two leaders who had overlapping responsibilities but different expectations, priorities and styles.
Prior to proposing the unified Human Foods Program, different divisions of the agency may have worked towards the same goal – or at times against each other – without adequate knowledge of other stakeholders’ actions, priorities or interpretations of issues
Bumpus told the Alliance for a Stronger FDA that a lack of transparency isn’t unique to FDA, but that in her current role she actively tried to reduce it.
“It’s a challenge for any institution, especially an institution where you have so many research projects ongoing,” she said, adding in her current position she was focused on putting “formal mechanisms in place to really data share, to method share, to understand what someone located in a different state, but still at the Agency, is working on and how you might collaborate with them.”